


Fresh, Unprocessed (Raw) Whole Milk: Safety, Health and Economic Issues
November 19, 2009Doodt rauwe melk ziektekiemen?
December 30, 2009A Response to Position Paper by Amanda Rose
By Ted Beals, MS, MD
Read this article in Dutch
Amanda Rose describes herself as a consumer and supporter of raw milk. She represented the “pro-raw milk view” at a symposium entitled “The Raw Milk Conundrum” sponsored by the American Veterinarian Medicine Association (AVMA) at their convention in Seattle, Washington, July 12, 2009. In her talk, she presented the results of an online survey she conducted, which asked a number of questions about consumer attitudes towards raw milk.
Shortly after the AVMA conference, Rose announced the publication of a position paper on raw milk entitled “Does raw milk kill pathogens? A visual analysis of the research on competitive exclusion” (http://rawmilkwhitepapers.com/assets/Does-raw-milk-kill-pathogens-12.pdf). In it she describes the notion that raw milk is a better pathogen fighter than pasteurized milk as an “urban legend.” She concludes: “The evidence suggests that we really cannot count on raw milk killing enough pathogens to ensure its safety.”
Evidence of Bias?
An important note about her online consumer survey, her presentation in Seattle and her position paper: she has framed the question to get the answer she apparently wants, namely that fresh raw milk does not kill pathogens. In her survey she asks respondants to make a judgement about the following statement: “The beneficial bacteria in raw milk kill the pathogenic bacteria.” Respondants are asked to judge whether this statement is absolute fiction, absolute truth or somewhere in between. The participants overwhelmingly answered that this statement was truth. It is my impression that Rose’s position paper was designed to correct that “misconception.”
Rose does not belong in the category of extremist fear mongers (she appears to reject the statement that drinking raw milk is “like playing Russian roulette with your health”). However, her position paper and pronouncements on the Internet place her in the category of those that wish to focus attention on the alleged dangers of drinking raw milk. Her position paper gives the impression that milk can contain extremely dangerous bacteria.
I do not disagree that milk, like all foods, can be contaminated with disease-causing microorganisms, but the inquiring public needs accurate and objective information. The opponents of raw milk have learned how best to scare people. Rose claims to provide balanced information, but this position paper is far from balanced; it is specifically styled to scare, not inform, the public. The best she has to say in favor of raw milk is a back-handed compliment—“the pleasure of consuming the food.” Saying that she drinks raw milk and might choose contaminated raw milk over contaminated commercial processed milk is not a balanced statement, nor one that provides consumers with the information they need to make informed choices about raw milk.
Competitive Exclusion
In the title of her position paper, Rose uses the phrase “competitive exclusion.” As a biologist, I prefer the phrase “competitive inhibition” as a more accurate description of this well recognized but complex biological phenomenon.
The term has been publicized by the probiotic industry. An accepted description is as follows: competitive exclusion (CE) is used to describe the process by which beneficial bacteria exclude bad bacteria or pathogens. CE implies the prevention of entry and establishment of a bacterial population into the gut. To succeed, the good bacteria must be better suited to establish or maintain itself in that gut environment. CE relates to the interactions of living bacteria colonies in mixed communities, where certain bacteria are able to inhibit (not exclude) others from becoming established. This phenomenon is not something that can be measured in test tubes. Rose inappropriately applies the term competitive exclusion to the ability of raw milk to kill off pathogens inoculated into laboratory samples of raw milk.
Those who look at the benefits and value of fresh, unprocessed whole milk (raw milk) point to competitive inhibition as only one of a variety of mechanisms that enable milk, fresh and unprocessed from the cow, to diminish the possibility that a virulent bacterial contaminant might colonize the gut and cause sickness. The lactoperoxidase system and lactoferrin are two of the many antibacterial enzyme systems that contribute to the competitive inhibition properties of fresh raw milk.
Scientific Papers
In her position paper, Rose focuses on seven scientific papers along with a document from the Food and Drug Administration, information from a private lab’s test results and unpublished preliminary information. A lot more on this subject can be found in the scientific literature, but I will focus on the reports that Rose cites in her paper.
Four of the papers look at what happens to pathogens when inoculated into milk.. Rose concludes that the weight of the evidence indicates that the pathogens are not killed, or are not adequately killed, to make the milk safe. She fails to inform her readers that in each of these papers, the results and conclusions of the authors is that the inoculated pathogens are killed.
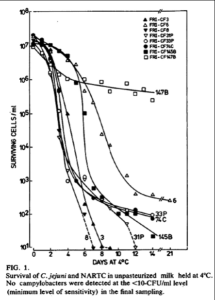
Rose first discusses “Prevalence and Survival of Campylobacter jejuni in Unpastuerized Milk” by Michael P. Doyle and Debra J. Roman (Applied and Environmental Microbiology, Nov 1982 44(5):1154-1158). In the paper, the authors provide a chart (above) showing the reduction in campylobacter—a reduction that can only be described as dramatic.
The authors inoculated raw milk with eight different strains of Campylobacter jejuni, shown by the different curves in this figure. Only three of these strains are from human sources (a fact Rose omits from her report). All strains do show reduction in pathogens with time. The only line without a steep decline (the steeper the line the faster the pathogens were dying) tracks a nonhuman strain.
In her paper, Rose created her own chart, which deemphasizes the extensive and dramatic results factually depicted by Doyle and Roman. That’s because the data she uses are from another experiment, reported in the same paper, which documents the fact that although the inoculated strains were dying, the generic bacteria already present in the raw milk were just as dramatically multiplying, increasing to as many as eight hundred million bacteria over the course of the experiment.
In another cited paper, researchers Massa, Goffredo, Altieri and Natola inoculated seven different strains of E. coli O157:H7 into fresh unprocessed whole milk to determine their fate after days of storage (Letters in Applied Microbiology 28(1):89-92). Like Doyle and Roman, they spiked the milk with extraordinarily high numbers of each pathogen (1,000,000 per ml—Doyle and Roman used 10,000,000 per ml). Even with these huge numbers of pathogens, the E. coli O157:H7 strains failed to grow and died off gradually. Actually, the purpose of this research was not to determine whether the pathogens were being killed, but whether it was acceptable to store milk at 8°C ( 46°F) rather than the standard 5° C (41° F). The authors conclude that the colder temperature should be used as the standard.
In the third paper, researchers Pitt, Harden and Hull used lower amounts of inocula of a different pathogen, Listeria monocytogenes, introduced into raw milk, but unlike the others they kept the milk at temperatures that optimize the growth of these bacteria (98.6° F) (Australian Journal of Dairy Technology 54(2):90-93). After fifty-six hours, no viable cells of L. mono were detectable. In a paper not cited by Rose, these same authors looked at other pathogens and concluded, “The growth of Staph. aureus, S. enteritidis, and L. monocytogenes in raw milk at 37° C was reduced markedly compared to the growth of these organisms in pasteurized milk” (Milchwissenschaft 2000 55(5):249-252).
In the fourth paper cited by Rose, Doyle working with Zhao and Wang looked at survival of E. coli O157:H7 at refrigerated and higher temperatures. They used a mixture of five research strains of this pathogen and concluded that E. coli O157:H7 did not grow at 5° C (41° F) and decreased over days (Journal of Food Protection 60(6):610-613).
Huge Numbers
It is important to understand that huge numbers of pathogens were added in these research protocols, because this practice makes it much easier to count the remaining bacteria.. What the general public needs to understand is that these are not conditions of our dairy practices. A simple calculation will make this point. When a person or a cow is having diarrheal sickness, their stool/feces contain extremely large numbers of the pathogen causing the disease. For the medical laboratories this makes it really easy to determine which bacteria is causing the diarrhea.
However, in dairy operations across this country, safety experts have been focusing on the fact that in some dairies a few of the cows that appear healthy and do not have intestinal disease with diarrhea, can be colonized by a pathogen and can shed the pathogen for short periods of time. But under these conditions, when pathogens appeared in the feces, their numbers were extremely low—in fact these pathogens could be called “background” pathogens compared to the large numbers of other (mostly beneficial) bacteria present.
So let us consider conditions closer to reality.. Suppose in the dairy herd there are some cows that are not sick, but are shedding small numbers of pathogens. If we wanted to contaminate some fresh unprocessed milk, how much manure would it take to equal the amounts used by the researchers that Rose cites in her position paper? There have been abundant publications on what happens to pathogens that exist in manure or dairy soil.
But these studies inoculate very large numbers of a pathogen in the material and then look at what happens to the numbers over time. There are far fewer articles that ask about the numbers of pathogens in the feces of farm animals, particularly when they are not clinically sick. Some of the best information is found in research projects that spike milk with pathogens. Before inoculating the milk, the researchers check to make sure that the cow’s feces are free from pathogens. On rare occasion they find that the cow is shedding one of the pathogens, and give the amount in their paper.
Although somewhat of a departure from reality, let us suppose we collected 1 gram of feces from one of those rare animals that is shedding a pathogen. That gram (about one thimbleful) will contain different amounts of pathogen (see below). Researchers have reported that feces with E. coli O157:H7, Campylobacter jejuni, or L. monocytogenes will contain about 500 cfu (colony forming units) in a gram.
To match the Doyle group’s inoculum you would need 20,000 thimblefuls of fresh manure from a cow shedding Campylobacter jejuni (10,000,000 cfu divided by 500 cfu/g). The inoculum from the Massa study was equivalent to 2,000 thimblefuls of manure from a cow shedding E. coli O157:H7 (1,000,000 cfu divided by 500 cfu/g). The inoculum from a study by Gaya, Medina and Nunez (Applied and Environmental Microbiology, Nov 1991 57(11):3355-3360), which inoculated raw milk with large amounts of L. monocytogenes,would equal only 20 thimblefuls from a cow shedding L. monocytogenes (10,000 cfu divided by 500 cfu/g).
Inoculums such as these may be standard procedure in a research laboratory, but they would never occur in a real dairy environment. That is a lot of manure! Even fractions of a thimbleful of manure would be conspicuous on the in-line filter. Besides, that filter would remove the material from the milk before it reached the storage tank. The most ardent proponent of raw milk would never suggest consuming milk containing such huge amounts of manure. Even the anti-microbial properties of raw milk can be overwhelmed!
BSK Challenge Test
Rose devoted a substantial portion of her paper to the “BSK Challenge Test.” This is not published research, but from the information I have from the laboratory that performed this testing, the researchers used a cocktail of three pathogens each in extremely large numbers (67,000,000 of Salmonella, 250,000,000 of E. coli O157:H7 and 220,000,000 of L. monocytogenes). They actually inoculated with only a portion of the final cocktail, so the amount of each pathogen added to the milk was less: 2,400,000 of Salmonella; 9,200,000 of E. coli O157:H7; and 8,100,000 of L. monocytogenes. We already have a visual idea of how much fresh manure it would take to match these amounts.
Furthermore, in this project all three pathogens were inoculated simultaneously into the milk sample. With some extremely large confined herds I have seen reports of two pathogens discovered together on the same farm, but never three. Even under these extremely unlikely laboratory conditions, the amounts of pathogens found in the milk after several days were reduced. Not only did the milk keep these three pathogens from growing, they were being killed. The lab report says the milk was maintained at 40° (I assume 40°F).
Five-Log Reduction
Rose makes an unsupported statement in her paper that “Microbiologists want to see a change of five logs in bacteria counts.” Such an assertion trivializes the complexity and controversy over what constitutes adequate destruction of pathogens. Some food safety publications talk about the desirability of a 5 log reduction of pathogens. But that is not the criteria used for milk. A threatened population of a pathogen does not die all at once. There is a gradual decrease, whether the killing is by heat or by biological antibacterial activity. Research shows the reductions essentially follow a logarithmic (base ten) pattern.
To date I know of no one who has proposed a criteria for adequate antibacterial effect in fresh raw milk. Any argument for adequacy of pathogen killing must consider the total number of pathogens present under real circumstances. A 5 log reduction is the same as saying 99.999% of the bacteria were killed. You could not measure a 5 log reduction starting with realistic numbers of bacteria because the procedures do not measure fractions of a bacterium—if you started with 2,000 bacteria, a 5 log reduction would be 0.02 bacterium which is the same as saying that if you performed the experiment 100 times, in two of those repeats there would be bacterium present. On the other hand if you started with 200,000,000 bacteria then a 5 log reduction would leave 2,000 bacteria. Wisely, none of the researchers in the papers Rose discusses mention her 5 log criteria. Nevertheless, Rose simplistically concludes that since none of their results demonstrated her arbitrary standard, then the milk did not kill enough pathogens.
A Legitimate Question
Rose then asks a legitimate question: are there enough pathogens in a glass of milk from any of these experiments to cause people to become sick? To help her readers visualize the amount that makes people sick, Amanda Rose draws bar graphs to show that on Day Six, most of the experimental milk in her selected publications will not have killed enough inoculated pathogens to drop below her red line (the FDA’s minimum infectious dose). But she fails to point out the incredible amounts of manure that would have to be shoveled into the farmer’s bulk tank to get the levels up to the inocula used in those laboratory studies.
One of the basic myths of the anti-raw milk folks is that it doesn’t make any difference how few pathogens get into the milk, because milk is such a nutrient-dense food, even one bacterium will rapidly multiply until everything is overwhelmed with bad bugs. All of the publications Amanda Rose selected demonstrate the fallacy of this scare tactic. Pathogens do not rapidly multiple in milk that is fresh and unpasteurized, they do not grow, in fact they die off.
Lactoperoxidase
Rose does admit that certain enzymes in raw milk can have antimicrobial effects. She discusses one study, by Althaus, Molina and Rodriguez (Journal of Dairy Science 84:1829- 1835), which looks at the lactoperoxidase system in ewe’s milk. Pathogens were not involved in this study. I believe the purpose for inclusion in her position paper was to show that in milk the amount of the enzyme active in this system varies. And since it varies, she argues that it can not be relied upon to make the milk safe. What Rose fails to understand is that the amount of the enzyme is not the limiting factor in this system, or in any enzyme system. The lactoperoxidase system is well recognized as a potent antibacterial system in fresh milk. In this system, depletion of the cofactors is what limits the antibacterial action.
The authors did not examine whether the variation in amount of enzyme would have any effect on the ability of the system to kill pathogens. They do conclude that the addition of the cofactors would make the system continue to be active at whatever levels of the enzyme were present in the milk. And since the specific cofactors in the lactoperoxidase (LP) system are abundantly available from beneficial bacteria in fresh unprocessed milk, the system appears to be fully active independent of the amount of the enzyme.
In the Gaya study, mentioned earlier the researchers concluded, “According to our results, the LP system exhibited a bactericidal activity against L. monocytogenes in raw milk at refrigeration temperatures” (Applied Environmental Microbiology 1991 57:3355- 3360).
Miscarriage
The remaining reference in the Rose paper concerns the ability of L. monocytogenes to enter different organs of the body (Bakardjiev AI and others. PLoS Pathogens 2(6):e66). Milk was not involved in this study. As background, it is helpful to realize that under usual conditions this particular pathogen exists and only grows inside of inflammatory cells that move around in our bodies. The research used pregnant laboratory guinea pigs. The pathogen was inoculated directly into the body and did not enter through the intestine, which is the normal foodborne route. Inflammatory cells containing multiplying pathogens moved through the body, and if there were enough of the pathogens (they were inoculating 7,500,000 bacteria into the animals), many organs became infected. The only reason that I can see that Rose discusses this research is to make the emotionally charged point that if the placenta became infected, an abortion ensues. As she comments: “The body then protects itself from the infection by expelling the baby and causing a miscarriage, stillbirth, or premature birth depending on the stage of the pregnancy.” It should be pointed out that this is not anything the authors said. They didn’t even study different stages of pregnancy.
Rose does correctly report the authors’ statement that a single bacterium is able to cause infection in the placenta, but her emotional message fails to tell her readers that the minimum infectious dose of foodborne L. monocytogenes is not one bacterium.. The FDA in their Bad Bug Book says that the infectious dose is unknown but states, “it is safe to assume that in susceptible persons, fewer than 1,000 total organisms may cause disease.”
The initial barrier to infection from this pathogen in milk is the wall of the intestine. Rose also conveniently fails to tell the readers that the authors found that the “placenta is relatively protected from infection.” Even with their direct injection into the body of the guinea pigs, the authors show that it takes large numbers circulating in the body to infect most organs, and greater numbers to overcome the unique protection afforded the placenta. However, they did show that once inside the placenta the pathogen flourishes and can cause abortion.
In summary, Rose describes her document as a visual analysis of the research on competitive exclusion. Her choice of studies, her misunderstanding of the science, the specific choice of data and her personal way of visualizing the information, coupled with emotionally charged statements, distort and exaggerate the researchers’ findings, obscuring the evidence showing that in fact, raw milk does kill off pathogens in realistic real-life situations. That fresh raw milk has the properties to kill pathogens is no urban legend; it is proven science.
Sidebars
More Than a Thimbleful!


The term cfu, or colony forming units, is a measure of bacterial colonies that grow individually on a culture plate in the laboratory. If spread widely enough, each colony would originate from a single speck of bacteria, multiplying until their numbers increase and they became visible. Technically, since it is possible for bacteria to clump together, several bacteria could clump to make one of those specks. For this reason the unit of measure is cfu, not numbers of bacteria.
Infectious Dose
In their Bad Bug Book, the FDA published infectious doses for the major pathogens. “Infectious dose” is shorthand for “minimum infectious dose,” which is meant to give an estimate of the fewest number of organisms that possibly were ingested by someone who became ill. In some older literature, a group of people were given a sample which contained various amounts of a pathogen, and researchers determined the infectious dose as the least amount that one person in the group ingested and had symptoms. Every student of infectious disease knows that this dose is not the amount that will make people sick. There are a whole list of factors that will change the amount necessary, but these factors all increase the number from the minimum. For example, it will take higher numbers to make a healthy person sick. Some research uses the amount that will cause half of the people to become sick as a useful yardstick. That number is a lot higher than the infectious dose used by the FDA. Unfortunately, that more reasonable standard has not been accepted by the public health agencies.
[include content_id=643]


6 Comments
Just as an aside… in the “worst” excess concentration study, it appears that it would have been necessary to have added almost 18,000 GALLONS of feces to the sample using typical contamination levels to obtain the sample contamination levels that were used!
And despite all this evidence if you do a quick search you will find stories of lots and lots of people getting very very sick from raw milk. How do you explain that, and how can you recommend with a clear conscience that people feed raw milk to babies whose immune systems are not developed enough to handle possible pathogens. Don’t get me wrong I lovi love raw milk and think it has great health benefits over store bought milk. But does that possible slight improvement in overall. Health warrant a possible bout with HUS THAT ccould be deadly or have severe lifelong consequences to the person, especially a baby? A more middle of the road approach recommendation I believe would be yes raw milk is great but be cautious about giving it to children under 5, the elderly, and those with deficient immune systems.
Sally Fallon Morell replies: There have been about 28 cases of HUS associated (associated, not proven) with raw milk during the past 10 years. There are 7,500 cases of HUS per year, the vast majority not in any way associated with raw milk but caused by other foods. Should these be banned also?
Many babies who are failing to thrive have their lives saved by raw milk. Children are the very ones who need raw milk the most. Raw milk provides powerful protection against asthma, which kills 9 people per day in the US–many of them children.
We have the technology today to ensure that all raw milk is safe–if health officials would stop opposing it, and join with us to have reasonable standards, we could reduce the incidence of HUS associated with raw milk to close to zero. These technologies involve rapid testing on the farm that is accurate, easy and inexpensive.
Until about 4 years ago I had significant problems with my immune system, something that my immunologist said couldn’t be fixed. At the end of 4 month viral infection I started to get symptoms of ME. I learnt that people with ME had been ‘cured’ when as cancer patients, they had their immune system suppressed. The idea suggested was that ME was caused by the immune system attacking itself or something else. Thus no identifiable virus, but all the fatigue and fever. I read that asthma patients had been cured with raw milk. I put 2 + 2 together and came up with the idea that raw milk might help me. I drank it for about two years. Amazing difference. I hardly ever get a cold now and if I do it goes very quickly. Prior to this I used to get more illnesses and have them for longer than anybody else. Now it’s the other way around. Of course it’s possible to get sick, but I buy my stuff from a safe source and I cant speak of it highly enough.
This Harvard debate I think raises questions about the stance people and organisations take with raw milk https://www.youtube.com/watch?v=iLRdihFi6gw.
Pete
Yes a “spokes person” such as Amanda is certainly NOT what we need.
Leaving out vitally important information and miss-representing information is a disgusting practice.
I notice that the CDC is now really distorting the issue of raw vs. pasteurized milk. They are very careful not to point out that the disease and death rate for pasteurized milk is higher than for raw milk.
The 2 deaths that the CDC attributed to raw milk in California (over an 11 year period) were not from raw milk but rather from a cheese made from raw milk that is not even legal in California. Political influence? Naw! Tell me it isn’t so!
You are quite right Jenny. People are a great deal more likely to get sick from eating lettuce or poultry! My own dairy farmer has celiac disease and drinks raw milk every day but will get ‘deathly’ sick if he drinks pasteurized milk!